Epidiolex: Uses, Dosage and Side effects

- Epidiolex
- 17 Aug 2023
Overview
What is Epidiolex?
Epidiolex is a prescription drug that is produced from cannabidiol (CBD), which is derived from the cannabis plant, popularly known as marijuana. Epidiolex is the first FDA-approved marijuana-derived medication.1Overview| Researched based study from Fda.gov Epidiolex is not a synthetic medication. Regulatory authorities have approved Epidiolex for treating specific seizures linked with certain medical disorders.
This article aims to provide an in-depth overview of Epidiolex, including its mode of action, uses, dosage, administration, storage, side effects, precautions, risks, and potential drug interactions.
How does Epidiolex work?
- Cannabidiol, the substance that makes up Epidiolex active, is thought to interact with several brain receptors, including the receptors for cannabinoids CB1 and CB2.2Overview| Researched based study from Nlm.nih.gov
- Epidiolex is considered to produce anticonvulsant effects by regulating these receptors, lowering the frequency and severity of seizures in certain epilepsy disorders.
- CBD may also affect other neurotransmitter systems to support its anti-inflammatory and neurotransmitter-modulating properties further.
Uses
What is the drug Epidiolex used for?
Epidiolex is primarily used to treat severe kinds of childhood epilepsy in individuals one year and older 3Uses| Researched based study from Epidiolex.com :
- Lennox-Gastaut syndrome (LGS).
- Dravet syndrome (DS).
- Tuberous sclerosis complex (TSC).
Lennox-Gastaut syndrome
- Lennox-Gastaut syndrome is severe epilepsy that appears in early childhood, usually before age four. 4Uses| Researched based study from Ninds.nih.gov
Dravet syndrome
- Dravet syndrome is a type of epilepsy that occurs in infancy or early childhood and can cause a wide range of symptoms from moderate to severe.5Uses| Researched based study from Ninds.nih.gov
Tuberous sclerosis complex
- A rare genetic illness causes non-cancerous tumors to grow in the brain and other body parts, including the spinal cord, eyes, heart, peripheral nerves, kidneys, lungs, and skin. 6Uses| Researched based study from Ninds.nih.gov
Multiple, hard-to-control seizures that frequently last into adulthood are the hallmark of these diseases. In individuals with these conditions, Epidiolex has demonstrated good effects in lowering the frequency of seizures, which improves their general quality of life.
Dosage
Epidiolex dosage
The dosage of Epidiolex varies depending on the patient’s age, weight, and the specific epilepsy syndrome being treated.
Epidiolex dosage for Lennox-Gastaut syndrome and Dravet syndrome (DS)
- Starting dose – 2.5 mg/kg, taken by mouth twice daily.
- Maintenance dose – 5 mg/kg, taken by mouth twice daily.
- Maximum dose – is 10 mg/kg administered twice daily by mouth.
Epidiolex dosage for Tuberous sclerosis complex (TBS)
- Starting dose – 2.5 mg/kg taken by mouth twice daily.
- Maintenance dose – 12.5 mg/kg taken by mouth twice daily.
- Maximum dose – 12.5 mg/kg twice daily.7Dosage| Researched based study from Fda.gov
The dosage may be altered by the doctor based on how well the child’s body tolerates the drug. Following the prescribing physician’s instructions carefully and maintaining regular medical supervision throughout the treatment period is crucial.
Administration
- The medicine is available as a 100 mg/ml oral solution.7Dosage| Researched based study from Fda.gov
- For measuring the solution, use the oral syringe provided with the drug.
- Never measure your medication with a regular spoon.
- It is usually administered orally to children.
- Epidiolex should be taken with meals and at the same time every day.8Dosage| Researched based study from Nlm.nih.gov
- To administer the full dose of Epidiolex, gently press the oral syringe’s plunger in the direction of the inside of the cheek.9Dosage| Researched based study from Epidiolex.com
- Do not force the plunger or aim the drug at the throat or back of the mouth to avoid choking.
- Before using the oral syringe, it should be rinsed in soapy water and air dried.
- A feeding tube might also be used to give the oral solution, and caregivers can consult a doctor about it and follow the instructions strictly.
Storage
Storage of Epidiolex
- Epidiolex should be stored at room temperature, between 20°C and 25°C (68°F and 77°F).
- It needs to be protected from sunlight, kept in the original packing, and fastened tightly.
- To avoid unintentional ingestion, Epidiolex must be stored away from children.
- As soon as the bottle of Epidiolex is opened, use it within 12 weeks.9Storage| Researched based study from Epidiolex.com
Side effects
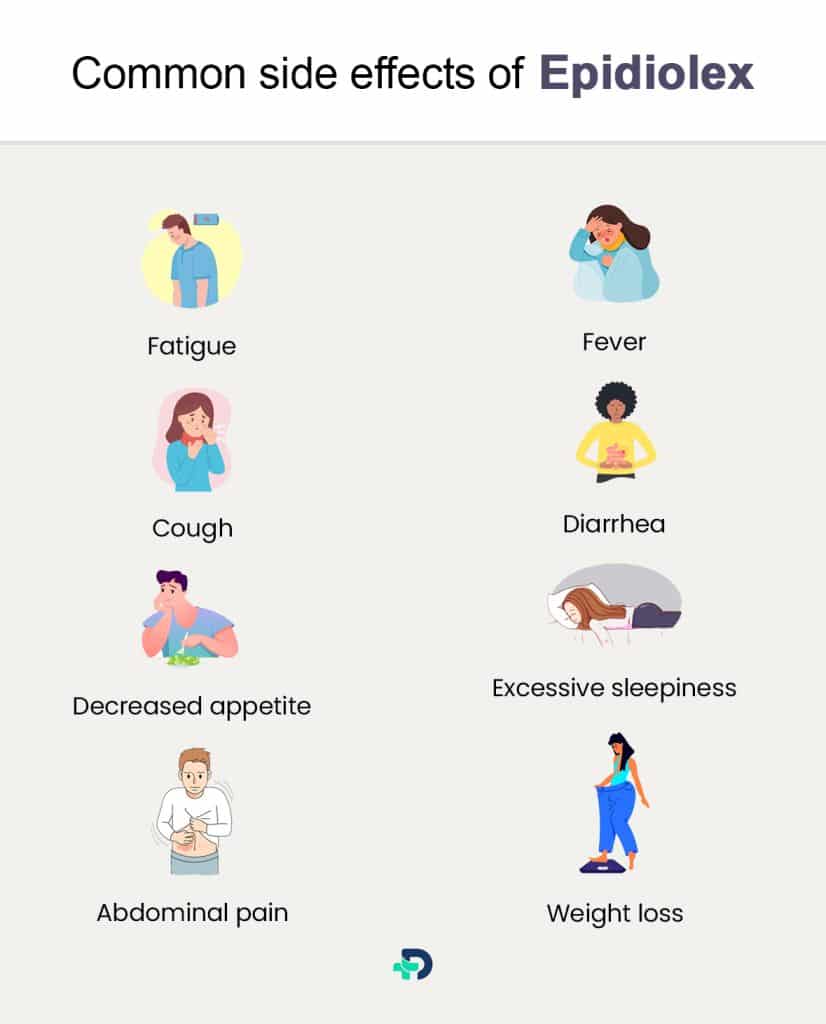
Like any medication, Epidiolex can cause side effects, although they tend to be mild to moderate in severity.
Common side effects reported by patients may include:
- Fatigue
- Fever
- Cough
- Diarrhea
- Feeling sick
- Decreased appetite
- Excessive sleepiness 10Side effects| Researched based study from Medlineplus.gov
- Poor sleep quality
- Abdominal pain
- Aggression
- Lethargy
- Pneumonia
- Skin rashes
- Weight loss
- Breathlessness
- Dark-colored urine
- Yellow skin or eyes
Risks
Potential Risks of Epidiolex
- Hypersensitivity – Epidiolex must be avoided in patients who have a history of allergies or hypersensitivity to cannabidiol or any of the product’s constituents.7Risks| Researched based study from Fda.gov
- Liver toxicity – Epidiolex may cause an increase in liver transaminases such as alanine aminotransferase (ALT) and aspartate aminotransferase (AST). 11Risks| Researched based study from Dailymed.Nlm.nih.gov If given in patients with liver disorders, it could worsen the condition.
- Suicidal thoughts or behavior – are increased in people using antiepileptic medicines, including Epidiolex, for any indication.11Risks| Researched based study from Dailymed.Nlm.nih.gov It should be avoided in people with depression or suicidal behavior.
- Drug dependence – Epidiolex should not be used in patients with a history of addiction to alcohol or any other substance. To reduce the danger of increasing seizure frequency and status epilepticus, Epidiolex should be tapered off gradually.
Precautions
Precautions
- In a hypersensitive reaction, the medicine should be discontinued immediately, and rapid medical treatment should be sought.
- The FDA warns pregnant women and nursing moms not to use cannabidiol (CBD), tetrahydrocannabinol (THC), or marijuana in any form.12Precautions| Researched based study from Fda.gov
- Monitoring transaminases like ALT and AST before and throughout Epidiolex treatment is critical because they can induce liver damage.
- Patients with unexplained nausea, right upper abdomen pain, nausea, vomiting, weight loss, problems with eating, exhaustion, yellow skin or eyes, or black urine should consult a healthcare physician as soon as possible.
- Patients using Epidiolex should be watched for drowsiness and should only drive or operate machinery, particularly motor vehicles, once they are reasonably assured that Epidiolex does not impair their vision, ability to think, make decisions, or perform motor skills.
- Suicidal thoughts and behaviors that may emerge during treatment should be warned about, and patients and caregivers should be alert for an increase or worsening of depression-related symptoms, any unusual shifts in behavior or mood, or the rise of suicidal ideas, behavior, or ideas of self-harm.
- Patients on Epidiolex should not stop taking it without first discussing it with their doctor.
Interactions
Drug interactions of Epidiolex
Epidiolex may interact with the following drugs and substances:
- Clobazam – a regularly prescribed antiepileptic medicine, when used with Epidiolex, can raise clobazam levels in the blood and increase the likelihood of specific clobazam side effects.11Interactions| Researched based study from Dailymed.Nlm.nih.gov
- Valproate – When used with Epidiolex, valproate may raise the risk of liver issues by boosting transaminase levels. Dose changes may be necessary.11Interactions| Researched based study from Dailymed.Nlm.nih.gov
- Alcohol – Using Epidiolex and other CNS depressants, including alcohol, may enhance the risk of drowsiness and sleepiness.7Interactions| Researched based study from Fda.gov
Before starting Epidiolex, it is recommended that people discuss all the medications they are currently taking, including prescription drugs, over-the-counter medicines, herbal drugs, and supplements, as the above list of drug interactions does not cover all the medications known to interact with Epidiolex. They will give customized guidance depending on an individual’s condition and medical requirements.
Any feedback on this article?
 This Articles content was accurate
This Articles content was accurate Very Informative Article
Very Informative Article I have a question or a comment
I have a question or a comment
 This article contains inaccurate content
This article contains inaccurate content This article was not helpful
This article was not helpful I have a question or a comment
I have a question or a comment
We appreciate your helpful feedback!
Checkout our social pages
References
-
U.S FOOD AND DRUG ADMINISTRATION
FDA Approves First Drug Comprised of an Active Ingredient Derived from Marijuana to Treat Rare, Severe Forms of Epilepsy | Overview
-
National Library of Medicine
Cannabidiol (CBD) | Overview
-
Jazz Pharmaceuticals
Starting on EPIDIOLEX | Uses
-
National Institute of Neurological Disorders and Stroke
Lennox-Gastaut Syndrome | Uses
-
National Institute of Neurological Disorders and Stroke
Dravet Syndrome | Uses
-
National Institute of Neurological Disorders and Stroke
Tuberous Sclerosis Complex | Uses
-
U.S FOOD AND DRUG ADMINISTARTION
EPIDIOLEX® (cannabidiol) oral solution | Dosage
-
National Library of Medicine
Epidiolex (Cannabidiol) Primer: Frequently Asked Questions for Patients and Caregivers | Dosage
-
Jazz Pharmaceuticals
EPIDIOLEX | Dosage
-
Medline Plus
Cannabidiol | Side effects
-
Daily Med
EPIDIOLEX- cannabidiol solution | Risks
-
U.S FOOD & DRUG ADMINISTRATION
What You Should Know About Using Cannabis, Including CBD, When Pregnant or Breastfeeding | Precautions