Aristada : What do I need to know?

- Aristada
- 16 Aug 2023
Overview
About Aristada
Aripiprazole lauroxil or Aristada was granted approval by the Food and Drug Administration (FDA) of the United States for treating schizophrenia among adult persons.2Overview| Researched based study from Nlm.nih.gov It has not been determined whether or whether aripiprazole lauroxil is effective in the treatment of dementia-related psychosis in individuals.
The long-acting injectable known as aripiprazole lauroxil can either be given once every four weeks or once every six weeks, taking into consideration the dose and the site of the injection. Aripiprazole lauroxil (ARISTADA) is an alternate medication to aripiprazole (MAINTENA).

Uses
What is Aristada used for?
In the management of schizophrenia
- Aristada Initio, when used together with oral aripiprazole, is the form of medication that is recommended for initiating Aristada therapy for schizophrenia in adults.4Uses| Researched based study from Texas.gov
Treatment of mental distress
- A mental illness that causes disturbed or unusual thinking, loss of interest in life, and strong or inappropriate emotions can be treated with aripiprazole extended-release injection (brand names Abilify Maintena, Aristada, and Aristada Initio).
Bipolar disorder treatment
- This medication can also be used in combination with other aripiprazole preparations. Aripiprazole extended-release injectable, sold under the brand name Abilify Maintena, is also used for the continued treatment of persons who have the manic-depressive disorder, often known as bipolar disorder.5Uses| Researched based study from Medlineplus.gov
Dosage
Administration and dosage of Aristada
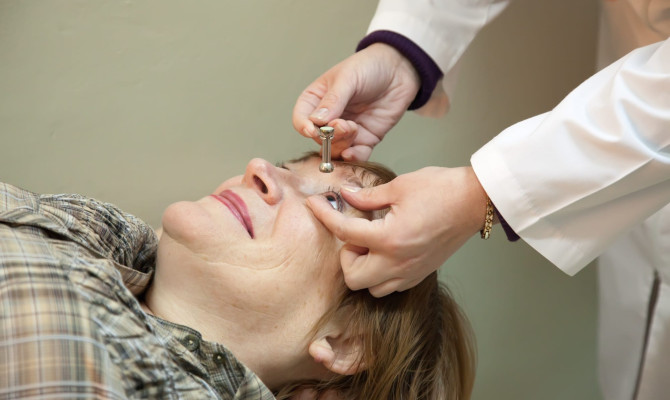
An intramuscular injection of aripiprazole lauroxil is given by a medical practitioner into either
- The deltoid muscle (only for the dose of 441.5 mg) or
- The gluteal muscle (for doses of 441.5 mg, 662 mg, or 882 mg)1Dosage| Researched based study from Fda.gov
Aripiprazole can be used in two different ways:
- As a single dose of either 441 mg, 662 mg, or 882 mg every four weeks
- A single dose of 882 mg every six weeks.1Dosage| Researched based study from Fda.gov
Patients who have never consumed aripiprazole must undergo a determination of whether or not a patient can tolerate oral aripiprazole before beginning treatment with aripiprazole lauroxil.
- In light of the fact that the half-life of oral aripiprazole is about 14 days, it is possible that a period of up to two weeks will be necessary to conduct an accurate evaluation of the patient’s response to aripiprazole.
- After the patient’s condition has been stabilized with oral aripiprazole, it is recommended that a dosage discussion take place.
- In conjunction with the initial injection of aripiprazole lauroxil, oral aripiprazole should be administered orally for a total of 21 days in a row.
- The extended-release injectable suspension of aripiprazole and lauroxil is offered in three different dosage strengths, including 441 mg in 1.6 mL, 662 mg in 2.4 mL, and 882 mg in 3.2 mL respectively.
- A sterile aqueous suspension of aripiprazole and lauroxil is contained in the prefilled syringe that is included in each package along with safety needles. Each syringe has a capacity of 5 milliliters.1Dosage| Researched based study from Fda.gov
How is Aristada administered?
ARISTADA can be used in two ways to start treatment:
- Option 1: Give one intramuscular injection of ARISTADA INITIO 675 mg and one dose of oral aripiprazole along with the first ARISTADA shot.
The first dose of ARISTADA can be given on the same day as ARISTADA INITIO or up to 10 days later.
Don’t put both ARISTADA INITIO and ARISTADA into the same shoulder or gluteal muscle at the same time. - Option 2: Give aripiprazole by mouth for 21 days in a row along with the first ARISTADA shot.
What is the mechanism of Aristada?
- The action mechanism of the prodrug aripiprazole lauroxil, which is derived from aripiprazole, is not known.3Dosage| Researched based study from Pbm.va.gov
- After being taken, aripiprazole lauroxil is converted to the active ingredient aripiprazole, which is a medicine that is available for purchase.
- It is speculated that the efficacy of aripiprazole is moderated by a mix of partial agonist activity at dopamine and 5-hydroxytryptamine 1A receptors and antagonist action at 5-HT2A receptors.
Side effects
Side effects of Aristada
Aripiprazole lauroxil or Aristada is associated with the following adverse reactions:
- Pain at the injection site
- Increased body weight
- Elevated blood creatinine phosphokinase
- Akathisia
- Headache
- Sleeplessness
- Restlessness.
- Tremors
- Cerebrovascular reactions like stroke
- Neuroleptic Malignant Syndrome
- Tardive Dyskinesia
- Alterations in one’s metabolic processes
- Compulsive behaviors, such as gambling, can become pathological.
- The onset of orthostatic hypotension
- Recurring falls
- Leukopenia
- Neutropenia
- Agranulocytosis
- Seizures
- Impairment of cognitive and motor capabilities
- Impaired body temperature control
- Dysphagia1Side effects| Researched based study from Fda.gov ,3Side effects| Researched based study from Pbm.va.gov
Contraindications
Contraindications of Aristada
- Increased risk of death in elderly patients suffering from Dementia.
- Patients who have a history of an adverse reaction to aripiprazole, such as anaphylaxis, should not take aripiprazole lauroxil.
- Aripiprazole can produce hypersensitivity reactions that range from pruritus and urticaria to anaphylaxis.4Contraindications| Researched based study from Texas.gov
Who should not take Aristada?
- People who have never taken ABILIFY®, ABILIFY MAINTENA®, or any other aripiprazole product in the past 1Contraindications| Researched based study from Fda.gov
- People who have or have had heart problems or a stroke
- People who have diabetes or high blood sugar;
- People who have a family history of diabetes
- High blood sugar patients
- Having or have had low or high blood pressure respectively 1Contraindications| Researched based study from Fda.gov
- Have or have had seizure activity (convulsions).
- Presently have or once have a low white blood cell count
- If you are pregnant or intend to become pregnant
- If you can suffer from complications if you receive an injection in the buttocks or arm
- If you are now breastfeeding or have future plans to do so.1Contraindications| Researched based study from Fda.gov
Your blood sugar should be checked before you start taking ARISTADA and periodically while you are receiving therapy with this medication.
Interactions
Interactions of Aristada with other medicines
Tell your doctor and pharmacist what prescription and over-the-counter medicines, vitamins, nutritional supplements, and herbal items you are taking or plan to take:
- Carbamazepine
- Clarithromycin
- Fluoxetine
- Itraconazole
- Ketoconazole
- Lorazepam
- Certain medications to control high blood pressure such as carvedilol, lisinopril, and prazosin
- Quinidine
- Rifampin
Your doctor may need to change the amount of medicine you take or keep a close eye on you to see if you have any side effects. There are a lot of other drugs that may interact with aripiprazole, so tell your doctor about all the drugs you are taking.
Overdose
Overdose of Aristada
Vomiting, somnolence, and tremor are some of the common side responses associated with taking an excessive amount of oral aripiprazole. Other clinically significant signs and symptoms include: 1Overdose| Researched based study from Fda.gov
- Acidosis
- Aggression
- An increase in aspartate aminotransferase
- Atrial fibrillation
- Slow heart rate
- Coma
- Confusion
- Convulsions
- Blood creatine phosphokinase increase
- Lowered consciousness
- Hyper- and hypo-tension
- Hypokalemia
- Tiredness
- Loss of consciousness
- QRS complex prolongation
- QT prolongation
- Pneumonia aspiration
- Respiratory arrest
- Seizures
Adverse effects
Adverse effects of Aristada
Cerebrovascular responses
- When compared with patients who got a placebo, elderly people diagnosed with dementia who were given aripiprazole, risperidone, and olanzapine had a greater frequency of cerebrovascular adverse effects, including fatalities.2Adverse effects| Researched based study from Nlm.nih.gov
Neuroleptic malignant syndrome (NMS)
- A potentially lethal symptom combination known as NMS is linked to the use of antipsychotic medications such as aripiprazole and lauroxil.
- After recovering from NMS, patients who need antipsychotic drugs should be monitored regularly.
Tardive dyskinesia
- A syndrome characterized by uncontrollable, dyskinetic movements that may or may not be reversible is known as tardive dyskinesia.
- With longer courses of treatment, there is a potential rise in the incidence of irreversible tardive dyskinesia.2Adverse effects| Researched based study from Nlm.nih.gov
- If signs of tardive dyskinesia begin to appear, it is recommended that the medication aripiprazole lauroxil be stopped immediately.
Alterations in metabolic function
- The metabolic conditions hyperglycemia, diabetes, dyslipidemia, and weight gain have all been linked to the use of atypical antipsychotic medications.
- 14% of patients in a long-term, open-label research of aripiprazole lauroxil in individuals with schizophrenia developed high HbA1c levels during the course of the study.2Adverse effects| Researched based study from Nlm.nih.gov
- These patients had normal glycated hemoglobin (Hb) A1c values at the beginning of the investigation.
Hypertension while standing or sitting
- It’s possible that aripiprazole could cause orthostatic hypotension.
- One patient out of one hundred and twenty receiving aripiprazole lauroxil reported experiencing orthostatic hypotension during a long-term study with an open-label.
Leukopenia, neutropenia, agranulocytosis
- Patients who have a previous history of leukopenia or neutropenia are encouraged to undergo frequent complete blood counts during the first few months of treatment.
- Aripiprazole lauroxil should be discontinued if severe neutropenia arises.
Seizures
- Patients who have a history of epileptic seizures or who have factors that reduce the seizure threshold should use aripiprazole lauroxil with extreme caution.
Possibility of cognitive and motor disability
- Aripiprazole lauroxil can have the same effect on a person’s judgment, reasoning, and motor skills that other antipsychotic medications do.
Monitoring internal temperature
- When prescribing aripiprazole lauroxil to patients who have conditions that may lead to an increase in the patient’s core body temperature, it is recommended that the patients be monitored appropriately.
Dysphagia
- Patients who are at risk for aspiration pneumonia should exercise extreme caution when taking aripiprazole lauroxil.
Takeaway
Key Takeaways
- Aristada is classified as an atypical antipsychotic medication and contains the active ingredient aripiprazole lauroxil. This class of drugs is known as atypical antipsychotics. Aristada is a drug that is sold under its own brand name. There isn’t a generic version of it that can be purchased right now.
- If you have schizophrenia, your doctor may give Aristada (aripiprazole lauroxil). Aristada is a medication that is available only with a doctor’s prescription and is normally administered to adult patients as part of a long-term treatment plan for schizophrenia.
- Before beginning therapy with Aristada, your physician may administer a single injection of a medicine called Aristada Initio. This medication is given as a single dose. The same active component can be found in both of these medications. However, only aristada is utilized as a treatment on a continuous basis. The medication will be given to the patient as an injection into a muscle by a qualified medical expert.
Any feedback on this article?
 This Articles content was accurate
This Articles content was accurate Very Informative Article
Very Informative Article I have a question or a comment
I have a question or a comment
 This article contains inaccurate content
This article contains inaccurate content This article was not helpful
This article was not helpful I have a question or a comment
I have a question or a comment
We appreciate your helpful feedback!
Checkout our social pages
References
-
U.S Food and Drug Administration
ARISTADA | Dosage | Adverse effects | Overdose | Contraindications
-
National Library of Medicine
Aripiprazole Lauroxil (Aristada): Long-Acting Atypical Antipsychotic Injection Approved for the Treatment of Patients with Schizophrenia | Overview | Precaution | Adverse effects
-
VA Pharmacy Benefits Management Services
Aripiprazole Long-acting Injection (Abilify Maintena, ARISTADA) | Dosage
-
TEXAS Health and Human services
Aripiprazole lauroxil | Uses | Contraindications
-
Medline Plus
Aripiprazole Injection | Uses | Interactions