Actemra: Uses, Side effects, and Interactions

- Actemra
- 16 Aug 2023
Overview
What is Actemra?
Tocilizumab, also known as Actemra, is a medication that suppresses the body’s immune response and is used to treat people with immune-related conditions and some cancers. A doctor will inject it directly into the vein or under the skin’s surface. This article explains some details about this medication, including its medical applications, side effects, precautions, and interactions.
Facts about Actemra
- Hoffmann-La Roche and Chugai developed Actemra in 2003.1Facts| Researched based study from Nlm.nih.gov
- The FDA, or Food and Drug Administration, of the United States authorized it in the first month of 2010 to manage medium to severe rheumatoid arthritis (RA).
- In June 2021, it was awarded a EUA, or emergency use authorization, for the administration of COVID-19 in the United States.2Facts| Researched based study from Fda.gov
- Tocilizumab belongs to the Interleukin-6 (IL-6) blocker class of medicines. 1Facts| Researched based study from Nlm.nih.gov
Mode of Action
What is the mode of action?
- Interleukin 6, also known as IL-6, is a cytokine involved in the immune system’s reaction and inflammatory responses in various disorders, including autoimmune conditions, multiple myeloma, and cancer of the prostate.3Mode of action| Researched based study from Nlm.nih.gov
- Tocilizumab binds to soluble and membrane-bound interleukin-6 receptors, preventing them from exerting pro-inflammatory actions.1Mode of action| Researched based study from Nlm.nih.gov
Uses
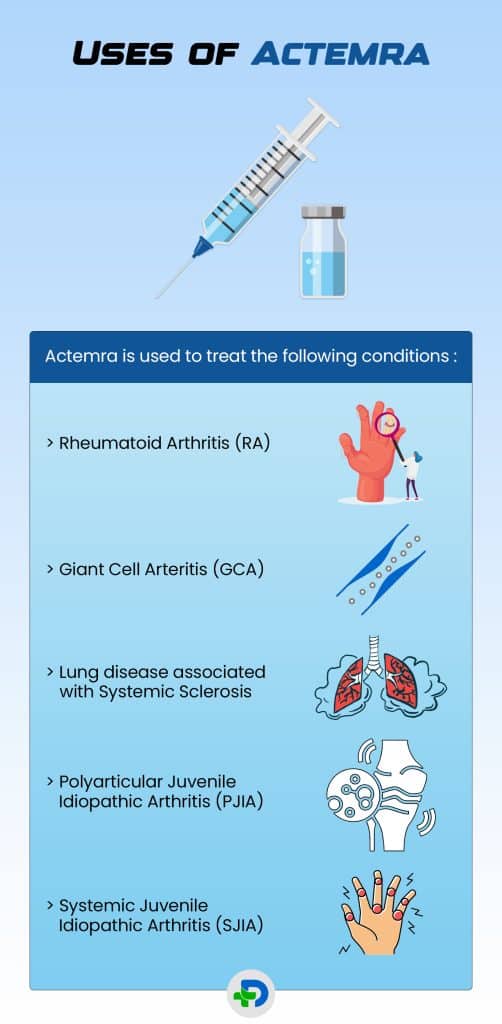
Uses of Actemra
Actemra is used to treat the following conditions
- Rheumatoid Arthritis (RA) – it is given to adult patients who have had unsatisfactory response to other anti-rheumatic drugs. 4Uses| Researched based study from Nlm.nih.gov
- Giant Cell Arteritis (GCA) in adults – can benefit from this medication since it lessens blood vessel enlargement and improves blood flow.
- Lung disease associated with Systemic Sclerosis – in adult patients, it delays the rate of deterioration in pulmonary function.
- Polyarticular Juvenile Idiopathic Arthritis (PJIA) – patients with juvenile idiopathic arthritis with polyarticular involvement who are two years of age or older can get better with tocilizumab.
- Systemic Juvenile Idiopathic Arthritis (SJIA) – patients aged two or more may benefit from the drug.
- Cytokine Release Syndrome (CRS) is a severe and potentially fatal reaction in adults and children aged two years and older after receiving specific immunotherapy treatments.5Uses| Researched based study from Medlineplus.gov
- COVID-19 – FDA has approved its usage in adult patients hospitalized with COVID-19 under oxygen supplementation, corticosteroids, ventilatory support, or extracorporeal membrane oxygenation (ECMO).6Uses| Researched based study from Nlm.nih.gov
Side effects
Side effects of Actemra
Some of the adverse effects of taking Actemra may include
- Increased infection risk.
- A sore throat.
- A stuffy or runny nose.
- Sneezing or coughing.
- Injection site pain, redness, and irritation.
- Headache.
- Ulcers in the mouth.
- High fever and chills.7Side effects| Researched based study from Rheumatology.org
- Distorted vision.
- Stools in black and tarry.
- Pain or tightness in the chest.
- Hypertension (high blood pressure).
- Bruising or bleeding that is unusual.
- Excessive fatigue or weakness.
- Difficult, painful, or scorching urinating.
- The body’s trunk is covered in painful blisters.
- Puffiness around the eyes, or on the face.
- Allergic symptoms include rashes, cheeks, eyelids, lips, or tongue swelling, and difficulties breathing.8Side effects| Researched based study from Hopkinsarthritis.org
- A liver injury may be indicated by dark yellow or brown urine, general malaise, loss of appetite, right upper tummy pain, unusual weakness or exhaustion, or yellowing of the eyes or skin.
- Breathing difficulties, lightheadedness, falls, and feeling especially weak or exhausted may indicate a low blood cell count.
- Hand or foot pain, tingling, or numbness.
- Fever, stomach pain, and a rapid change in bowel habits are all symptoms of stomach or intestinal tears.
- Rarely Anaphylaxis – rash, itching, hoarseness, difficulty breathing, difficulty swallowing, or swollen face, mouth, or hands.
Anyone exhibiting any symptom, whether one of the side effects listed above or not, should consult their doctor immediately.
Risk
What are the associated risk of Actemra?
This drug raises the likelihood of having the following issues
- Tocilizumab may make a person more susceptible to infections or worsen existing diseases.
- Patients may acquire extensive infections caused by fungi (e.g., candidiasis, aspergillosis, pneumocystis).9Risk| Researched based study from Nlm.nih.gov
- Opportunistic pathogens cause bacterial, viral, and other diseases.
- The risk of some cancers may arise due to the drug’s immune system-altering properties. The patients should report swollen lymph nodes in the neck area, underarms, or groin, unexplained weight loss, unexpected bleeding, bruises, or weakness to their doctor.
- It decreases the number of specific blood cells in the body. As a result, one might start bleeding or become infected more easily.
- It can potentially cause significant stomach and intestinal problems, especially if the patient has a history of ulceration or diverticulosis.
- Tocilizumab administration raises the risk of liver injury.
- This medication may raise cholesterol and fat levels in the blood.
- Tocilizumab may induce headaches and skin problems such as a rash or itching when administered or within 24 hours of receiving it.
- This medication may induce severe allergic responses, including Anaphylaxis, which can be fatal and require rapid medical intervention.
Precautions
Who should not use Actemra?
- Should not be used in people who have experienced hypersensitivity reactions to bacteria. 10Precautions| Researched based study from Nlm.nih.gov
- People should tell their doctor if they are taking other medications for any condition.
- Before treatment check for latent Tuberculosis (TB).
- Consider the risks and advantages of treatment before starting it in individuals with chronic or recurring infections.
- Medical professionals should monitor the individual’s platelets, neutrophils, ALT, and AST plasma concentrations, as well as any symptoms of infection, such as tuberculosis or candidiasis.10Precautions| Researched based study from Nlm.nih.gov
- The doctor should stop the therapy if a significant infection appears until it has been contained.
- Avoid receiving any immunizations with live viruses if you’re on tocilizumab.11Precautions| Researched based study from repository.phb.ac.idAlso stay away from people who has just received live virus immunizations. Example, flu vaccine.
- Only if the benefits outweigh the risks to the mother and the developing baby may bacteria be used during pregnancy.
- The impact of this medication on a breastfeeding newborn is uncertain, as is whether it goes into breast milk. Before you begin breastfeeding, consult your doctor.
- Do not take tocilizumab if you have another active infection.
- Treatment with this drug is not advised for patients whose increased ALT or AST levels are more than ten times the maximum level of the guideline ranges.
- Avoid contact with those who have diseases that could transmit to others. For instance, chickenpox, TB, and influenza.
- Avoid being around ill or infected persons to prevent infection. Wash your hands frequently.
- Gently floss and brush your teeth daily to avoid dental infections.
- To avoid bleeding and infections, employ caution when using sharp tools like blades, razors, or nail clippers.
- Other medications, including prescription and over-the-counter drugs and herbal and vitamin supplements, should not be taken unless discussed with a doctor.
Interactions
Interactions of Actemra with other medicines
Tocilizumab may alter the way drugs operate or increase the likelihood of significant adverse effects
- Other immunosuppressants, such as methotrexate and corticosteroids, can lower a person’s immunity and make them vulnerable to severe and life-threatening infections.12Interactions| Researched based study from Clevelandclinic.org
- This medication may hasten the clearance of other drugs from the human body and may change how they work. Some cholesterol medications, for example, atorvastatin, lovastatin, and simvastatin.12Interactions| Researched based study from Clevelandclinic.org
- It may reduce the efficacy of birth control drugs such as tablets, patches, or rings and could result in pregnancy. 13Interactions| Researched based study from Fda.gov One should discuss taking extra effective birth control with their doctor while under this medicine.
- Do not use this medication with any live virus vaccines since it reduces immunity, and the virus in the vaccine may end up causing infection rather than developing immunity. 14Interactions| Researched based study from Fda.gov
- This medication may interact with omeprazole, cyclosporin, and warfarin.1Interactions| Researched based study from Nlm.nih.gov
- Additionally, this drug could react with biologic medicines like rituximab, certolizumab, etanercept, and abatacept.15Interactions| Researched based study from Nlm.nih.gov
Takeaway
Takeaway
Before using tocilizumab, an individual should keep a list of the products they use, including prescription and nonprescription drugs, and then discuss it with their healthcare provider and pharmacist. Inform doctors that you smoke, consume alcoholic beverages, or use illegal substances. Certain products may have an interaction with the medication. Without consulting with a physician, do not begin, halt, or adjust the dosage of any medicines.
Any feedback on this article?
 This Articles content was accurate
This Articles content was accurate Very Informative Article
Very Informative Article I have a question or a comment
I have a question or a comment
 This article contains inaccurate content
This article contains inaccurate content This article was not helpful
This article was not helpful I have a question or a comment
I have a question or a comment
We appreciate your helpful feedback!
Checkout our social pages
References
-
National Library of Medicine
Tocilizumab: A new anti-rheumatic drug | Facts
-
U.S FOOD & DRUG Administration
Coronavirus (COVID-19) Update: FDA Authorizes Drug for Treatment of COVID-19 | Facts
-
National Library of Medicine
Interleukin-6 and chronic inflammation | Mode of Action
-
National Library of Medicine
Tocilizumab in rheumatoid arthritis: efficacy, safety and its place in therapy | Uses
-
MedlinePlus
Tocilizumab Injection | Uses
-
National Library of Medicine
Tocilizumab for treatment patients with COVID-19: Recommended medication for novel disease | Uses
-
American College of Rheumatology
Tocilizumab (Actemra) | Side effects
-
Johns Hopkins Arthritis Center
Tocilizumab (Actemra®) | Side effects
-
National Library of Medicine
Invasive pulmonary aspergillosis after treatment with tocilizumab in a patient with COVID-19 ARDS: a case report | Risk
-
National Library of Medicine
Tocilizumab | Precautions
-
Elsevier
Tocilizumab | Precautions
-
Cleveland Clinic
Tocilizumab Injection | Interactions
-
U.S FOOD & DRUG Administration
ACTEMRA® (tocilizumab) | Interactions
-
U.S FOOD & DRUG Administration
FACT SHEET FOR HEALTHCARE PROVIDERS: EMERGENCY USE AUTHORIZATION FOR ACTEMRA® | Interactions
-
National Library of Medicine
Clinical efficacy of abatacept, tocilizumab, and etanercept in Japanese rheumatoid arthritis patients with inadequate response to anti-TNF monoclonal antibodies | Interactions